Analysis of Fenton Process and Combined Systems On Leachate Treatment and Control in Landfills
Contents [show]
Summary
Fenton process is an advanced oxidation method used in the breakdown of organic and inorganic contaminants in landfill leachate. However, leachate composition in landfills varies significantly around the world. This review describes and draws conclusions from the pilot studies performed in Spain, China, Estonia, and the United States of America using the Fenton methodology in combination with other advanced treatment processes. The Fenton process possesses various advantages in leachate treatment and control and is commonly used in landfill treatment around the world, however, there are some limitations to this process, which can be significantly overcomed when combined with other modes of treatment due to potential synergistic effects.
Chapter 1: Introduction to Landfill and Leachate
One of the biggest problems associated with our growing population and consumption, is the disposal of our municipal solid waste (MSW) globally (Vergara & Tchobanoglous, 2012). The generation of waste contributes to the contamination of our soil and water, which in return, adversely impacts our health. Significant volume of waste is now facing capacity challenges, thus, finding the most efficient and economical strategy for waste disposal is an important ongoing research today.
About thirty years ago, landfills became the most popular source of the disposition of MSW. This method stores waste in a large, excavated area and covers it with various geosynthetic layers and impermeable soil to avoid contact with its surroundings. The system is also engineered away from environmentally sensitive areas and monitored daily to assure its compliance with the environmental regulations. However, even though landfilling provides a solution to our waste problem, it is important to note that the complexity and sensitivity of landfill treatment requires highly-skilled labour, especially when operation and maintenance require complicated equipment and processes (Yidong, Xin, Shuai, & Ancheng, 2012).
One major concern is how landfills respond to the percolation of water through precipitation, evapotranspiration, surface/agricultural runoff, infiltration and groundwater intrusion (Costa, Alfaia, & Campos, 2019). When water is exposed to the landfill, it becomes leachate, which is a highly concentrated effluent that consists of organic and inorganic matters like humic acids, ammonia, nitrogen, heavy metals, xenobiotics and inorganic salts (Primo, Rueda, Rivero, & Ortiz, 2008).
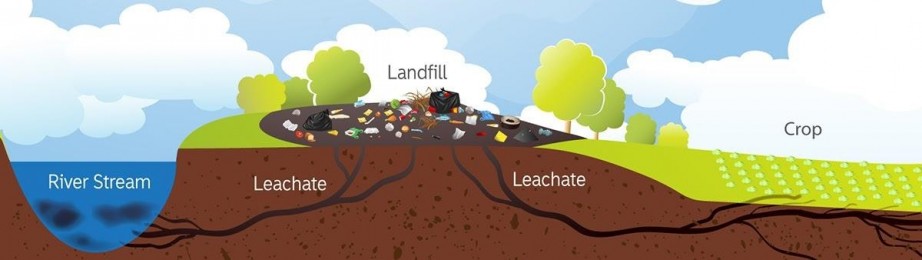
Figure 1.1 shows a possible contamination of leachate from a landfill. The effluent can contaminate its surroundings in all possible pathways such as the river stream, ground water and crops. Consequently, this scenario poses various health risks, thus, inspection of wastewater discharge monitoring of landfills is crucial.

Table 1.1 shows the average variation of leachate in Northern America over time. Landfill leachate is dependent on parameters such as pH level, biochemical oxygen demand (BOD), chemical oxygen demand (COD), total Kjeldahl nitrogen (TKN), ammoniacal nitrogen (NH4+, +N), phosphorus, chloride, alkalinity, conductivity, total dissolved solids, and heavy metals. The composition cannot be similarly applied and assumed to leachate outside of this region as the composition is also affected by non-chemical factors such as climate and economic conditions (Costa, Alfaia, & Campos, 2019).
The results show that the age of landfill is a major factor that determines the characteristic of leachate. Mature landfills (>10 years) are stabilized and have independently reduced its biochemical and chemical concentrations. The rate at which these compounds degrade, however, is dependent on the properties of its location and quantity of volatile contaminants.
Chapter 2: Landfill Leachate Treatment
It is important to emphasize that once leachate touches another form of fluid, such as the groundwater, the other body also becomes leachate. Thus, treating leachate before it is discharged into surface disposals or sewers is a pivotal component of landfilling. Therefore, landfill sites must be equipped to collect, store, and treat leachate to the federal standards (Byrne, n.d.).
The classical treatment methods of removing the organic and inorganic substances in leachate is through biological processing (aerobic and anaerobic), chemical and physical treatment (flotation, coagulation/flocculation, chemical oxidation, ion exchange and electrochemical treatment) and followed by member filtration (microfiltration, ultrafiltration, nano-filtration, and reverse osmosis) (Byrne, n.d.). Usually, biological processing and chemical and physicochemical treatment are effective initial processes. However, this is only true when the biological oxygen demand to chemical oxygen demand ratio is high (BOD5/COD) or only in the non-stabilized early stages of landfills (
Figure 2.1 shows an overview of the classic method by which wastewater is treated. In the primary level, large particles are removed through prefiltration. In the secondary level, biological processes and physicochemical processes are applied to remove organic compounds and settleable solid. Lastly, advanced stages (not pictured) are necessary for treatment above the first and second phase. Clarifiers may be used as intermediate steps. Advanced oxidation processes fall in the advanced category, necessary when classic biological, physical, and/or chemical processes alone do not provide satisfactory results in meeting federal contaminant standards.

However, it is pertinent to note that sanitary landfill leachate is highly and complex polluted wastewater. Treatment procedures must consider the relatively small flow rates and the complex leachate composition which makes it different from sewage and other kinds of wastewater (Stegmann, Heyer, & Cossu, 2005).
An effective alternative to the lowering BOD5/COD ratio is using advanced oxidation processes (AOPs) to reduce the organic compounds (COD) of the leachate (Primo et al., 2008). This method is usually combined with conventional biological practices to achieve maximum retrieval of recalcitrant compounds. Several AOPs have been developed worldwide and have been proven to be effective, but with an additional level of treatment and a more advantageous approach, the cost of recalcitrants removal inevitably increase.Chapter 3: Overview of the Fenton Process
One of the most cost effective and widely used AOP is the Fenton Process. It was developed by Henry John Horstman Fenton and was named the Fenton’s reagent. Initially, it was designed as an oxidizer for tartaric acid and was not a part of wastewater treatment until the late 1960s. Today, it is used in several countries to enhance the oxidation process of recalcitrants in leachate.
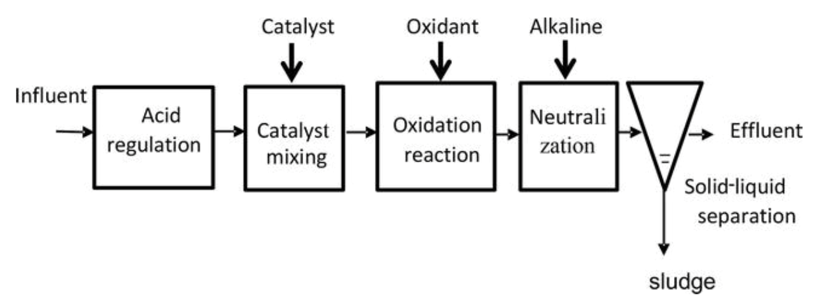
Figure 3.1 shows the continuous flow of the Fenton process. Firstly, the influent enters the system and acid is regulated. Then, ferrous iron is added to catalyze the core reaction. In the oxidation process, hydrogen peroxide is added to make radicals and oxidize organic compounds that are resistant to conventional treatment processes. In neutralization, alkaline is added to neutralize the pH level of the water. Lastly, solid sludge is separated from the treated water and the effluent is released into sewers of other surface disposal.
The organic compounds broken down in the leachate by the free radicals composes the COD (chemical oxygen demand). Thus, using the Fenton technique for oxidation is usually still paired with the conventional biological process. In the next chapter, the effect of the Fenton Process in COD concentration is further discussed to analyze the efficiency of the Fenton Process.
3.1 Chemical Reactions in the Fenton Process
In 1894, Fenton discovered that when ferrous iron (such as FeSO4, Fe2+) is combined with hydrogen peroxide (H2O2), it produces hydroxide ion (OH-) and hydroxyl free radicals (•OH) that has an oxidation potential of 2.8 V. In leachate treatment, these hydroxyl free radicals are effective oxidizers of the organic compounds and the iron complex produced are effective flocculants. Note that the iron is added in gradual amounts as the catalyst and the hydrogen peroxide is added continuously to produce the hydroxyl radicals. The chain of chemical reactions that is traditionally accepted as the Fenton degradation of compounds are as follows (Babuponnusami & Muthukumar, 2014).
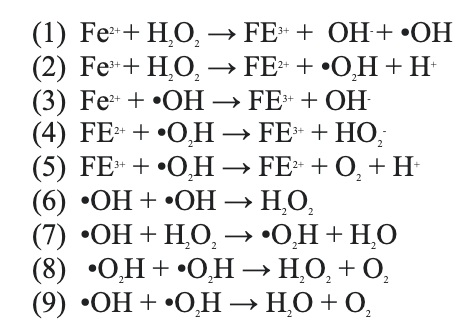
(1) The ferrous iron is combined with hydrogen peroxide to produce ferric ions, hydroxide and hydroxyl free radicals. This is known as the core of Fenton chemistry. (2) Ferric irons mix with hydrogen peroxide to produce more ferrous irons and more radicals. This is the Fenton like reaction that starts the cyclic ferrous iron regeneration. The hydroperoxyl formed (•O2H) is another free radical that can oxidize the organic compounds, but they are weaker than hydroxyl free radicals. (2)(3)(4)(5) Hydrogen peroxide is consumed and there is a cyclic mechanism of ferrous iron - ferric iron - ferrous iron. The end products are the ferrous ion, oxygen and hydrogen. (6)(7)(8)(9) Radical-radical reaction and hydrogen peroxide-radical reaction also occur in the Fenton process. The end products are water and oxygen.
This schematic shows the complexity of Fenton chemistry-- hydroxyl free radicals are formed in the first reaction but may get attracted by ferrous irons, hydrogen peroxide, hydroperoxyl radicals and/or another hydroxyl free radical. Thereby, hydrogen peroxide can make and break free radicals. In leachate treatment, the hydroxyl free radicals also act with organic compounds and break it down.
The following are potential reactions with R as radicals (Babuponnusami & Muthukumar, 2014):
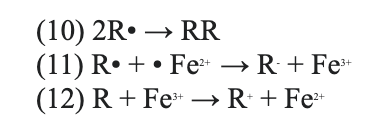
(10) Free radicals reaction with organics in the wastewater to form stable dimmers. (11)(12) Free radicals with ferrous or ferric iron react to break down organic compounds and have a solid ferric sludge by-product.
3.2 The Effect of pH Level on Hydroxyl Free Radicals
Although the Fenton process is an effective method of oxidation, one limiting factor is that it has to perform in a low pH level (Jung, Lim, Park, & Kim, 2009), typically 3. This is because of the hydrogen peroxide and iron speciation factors. In basic solutions, ferrous irons are inactive and hydrogen peroxides are likely to decompose. Moreover, the oxidation potential and capacity of the hydroxyl free radicals increases with decreasing pH.
However, complex iron species and stable oxonium ion [H3O2] + exists at very low pH values and this decreases the reactivity of ferrous iron and hydrogen peroxide. Therefore, having a low pH is both limiting and enhancing the Fenton process.
3.3 Advantages of the Fenton Process
The biggest advantage of the Fenton process is its high removal efficiency of organic compounds (Wang et al., 2012). It is widely used because the oxidation technology and operation produce short reaction time compared to other AOPs. The Fenton reagents, hydrogen peroxide and ferrous ions are also readily available, have easy storage, safe to handle and do not cause environmental damages. Additionally, there is no required activation energy to start the reaction and the process can be performed at room temperature and atmospheric pressure (Primo et al., 2008).
3.4 Disadvantages of the Fenton Process
On the other hand, the Fenton process’ main disadvantage is the high cost of the hydrogen peroxide and the significant amount of solid ferric sludge produced before discharge. Firstly, the oxidation through the hydroxyl free radical is still not performing at maximum efficiency. One major factor to this is the scavenging of the hydrogen peroxide during the chain reactions. Thus, even though it is part of the key compounds that initializes the Fenton chemistry, it also wastes the oxidants. Additionally, hydrogen peroxide decomposes not only at high pH, but also in the absence of organic compounds. Without another source to react with, hydrogen peroxides combine to make oxygen and water (Babuponnusami & Muthukumar, 2014). Next, although the Fenton reagents does not cause any environmental damages, its ferric sludge by-product does, which typically are dried and directed to landfills (Domingues, Gomes, Quina, Quinta-Ferreira, & Martins, 2018). The continuous loss of irons and sludge formation takes part in economic and environmental drawbacks. Thus, more research is required to understand the recovery or irons and its generation-degeneration cycle (Xu et al., 2020).
3.5 Advancements in the Fenton Process
Potential advancements in the Fenton Process are being considered mainly to improve oxidation efficiency or to reduce ferric sludge formation. Some enhanced Fenton Processes are the Photo-Fenton Process (Pouran, Aziz, & Daud, 2015), Electro Fenton Process (Alfaya, Iglesias, Pazos, & Sanromán, 2015), Sono-Fenton Process (Ranjit, Palanivelu, & Lee, 2008), use of modified iron source and the reuse of the iron-containing sludge. The Photo-Fenton Process uses UV radiation in the core of the Fenton chemistry to generate more hydroxyl free radicals. The Electro Fenton Process generates H2O2 in situ using two electron reductions of oxygen and two hydrogen atoms. Also, ferric ion is reduced to Fe2+ by electrochemical regeneration. Hydroxyl free irons can also be generated by a high oxygen overvoltage water oxidation of hydrogen peroxide. The Sono-Fenton Process uses ultrasound in the Fenton reagents to enhance oxidation rate by producing more free radicals. In using modified iron, iron salts are used as catalysts instead of ferrous ions. Lastly, the concept of reusing of iron in the solid sludge has increasing interest among researchers worldwide. Combining these techniques can lead to increase in efficiency of treatment and decrease in solid sludge by-products (Xu et al., 2020).
Chapter 4: Pilot Studies Involving Fenton Process Around the World
Optimal leachate treatment, to fully reduce the negative impact on the environment, is today’s challenge. However, the complexity of the leachate composition makes it very difficult to formulate general recommendations. Variations in leachates, in particular their variation both over time and from site to site, means that the most appropriate treatment should be simple, universal, and adaptable (Renou, Givaudan, Poulain, Dirassouyan, & Moulin, 2008). Major criteria such as initial leachate quality and final discharge limits imposed by local or municipal regulators should be thoroughly considered before executing leachate control and treatment plans.
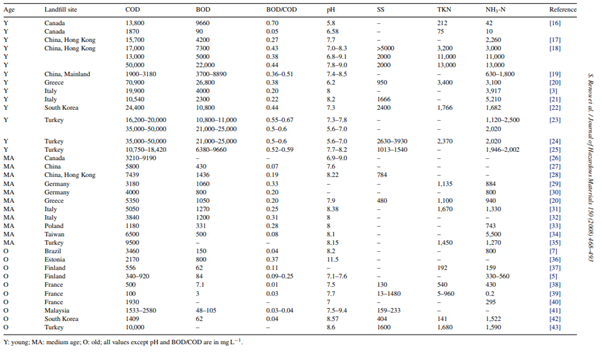
In landfills, different physical, chemical, and biological processes occur which, in turn, constantly affect the composition of the leachate, influencing its characteristics (Qasim & Chiang, 1994). Figure 4.1 summarizes the range of leachate composition in various landfill sites around the world. Standalone Fenton Process on landfill leachate has been observed to have COD removal efficiency of around 58% to 75% from research such as Trujillo, Font, and Sánchez (2006), Zazouli, Yousefi, Eslami, and Ardebilian (2012) and Ahmadian et al. (2013). While standalone Fenton process provides appreciable treatment of leachate, it suffers the drawbacks as evaluated in Chapter 3. This chapter summarises and evaluates pilot strategies around the world in combining Fenton process with additional treatment processes to advance the efficacy of leachate treatment in landfills, specifically pilot plants from Spain, China, Estonia, and United States of America.
4.1 Pilot Study in Cantabria, Spain
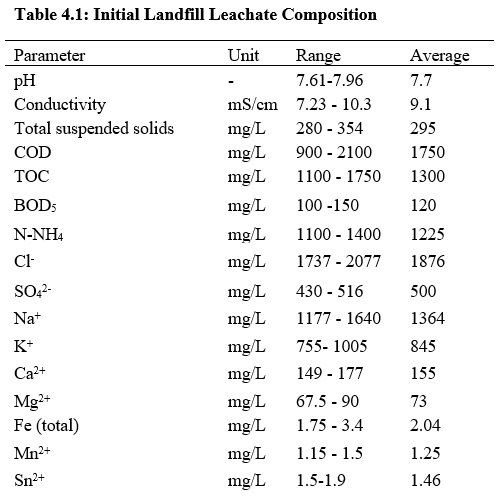
A pilot plant study done by Primo et al. (2008) was conducted to evaluate the viability of Fenton treatment as pre-treatment to increase the biodegradability of leachate or reduce the toxicity of the leachate in Spain. The leachate used in the study was collected from a municipal landfill in Cantabria, Spain. The treatment of leachate in the landfill was initially treated in situ by a biological process. Table 4.1 shows the initial landfill leachate composition in the landfill.
In the pilot plant, the landfill leachate treatment consisted of three steps: Fenton oxidation, neutralisation, and finally ultrafiltration with submerged membranes. The aim of the neutralization step was to precipitate and remove iron which was used as catalyst in the Fenton process, and the ultrafiltration step was to enhance the clarification and achieve lower turbidity.
4.1.1 Methodology
The leachate tank was connected to the Fenton’s reactor tank, neutralisation tank and ultrafiltration tank as depicted in Figure 4.2. Fenton reagents (H2O2 and Fe2+) were pumped into the reactor with H2SO4 (sulfuric acid) to provide acidic pH control of 3. In the neutralisation tank, CaOH2 (calcium hydroxide) or NaOH (sodium hydroxide) were pumped in to neutralise the effluent while ultrafiltration was carried out in the final tank using submerged membranes units.

4.1.2 Results
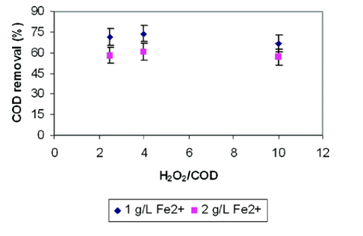
The efficiency of Fenton oxidation, expressed as COD, was investigated by varying the initial H2O2/COD weight ratio with Fe2+ concentration. Results depicted in Figure 4.3 shows how the Fenton reagents affected the COD removal. H2O2/COD ratios did not have a significant impact on COD removal, whereas COD removal shows notable decreases as Fe2+ concentration increases from 1g/L to 2g/L across all H2O2/COD ratios. This fact could be due to the presence of a refractory organic fraction that was not susceptible to Fenton oxidation, which are low molecular weight organic acids.
Optimum concentration ratios found in the experiments were: H2O2/COD = 3.3 and H2O2/Fe2+ = 7. A second Fenton process was conducted to evaluate the possibility of increasing COD removal with a second Fenton oxidation step. However, COD did not decrease considerably after a second Fenton reaction. Thus, the results obtained showed that after the first oxidation process, the remaining organic material was recalcitrant to secondary Fenton treatment process.
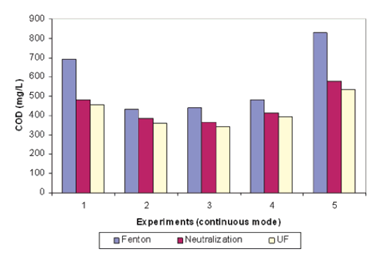
Neutralisation reaction performed to precipitate and remove iron registered low COD reduction of around 10% to 30% of Fenton effluent concentration depending on neutralisation reagent, with CaOH2 showing better reduction results due to its capability to remove more sulfate ions. Low efficiency could be attributed to low molecular weight oxidative by-products being more difficult to precipitate than high molecular weight compounds. A similar phenomenon was registered with the ultrafiltration step as remaining organic compounds were likely too small to be retained by the membrane. Figure 4.4 shows the COD concentration at each step of the treatment process across five different experimental runs.
Results from the pilot plant experiments show that the lowest COD concentration was approximately 400mg/L. Table 4.2 shows the percentage of COD, TOC and TSS removal after treatment. Nevertheless, such chemical oxidation processes using the Fenton process are not adequate in meeting requirements for disposal under Spanish legislation which requires a threshold COD value of 160mg/L.
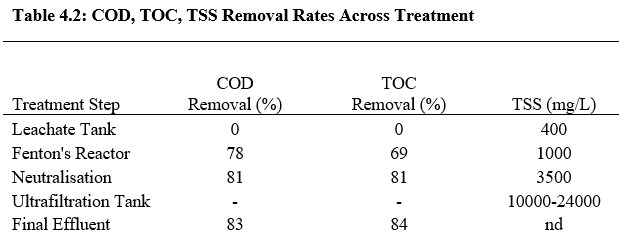
4.1.3 Discussion
This pilot plant study mainly considered COD, TOC and TSS removal. However, there are other remaining compounds such as ammonium and other inorganic salts that were not considered for adequacy and evaluated in this research. A total polishing of the effluent could be achieved by incorporation of a process with high efficiency in ammonium removal e.g., electrochemical oxidation, followed by a membrane unit, such as reverse osmosis, for the removal of inorganic salts.
4.2 Pilot Study in Zhejiang, China
A pilot plant study done by Wang et al. (2012) was conducted to consider advanced treatment of leachate using Fenton reaction Biological Anaerobic Filter (BANF) and biological aerated filter (BAF) in response to new discharge standard in China. BAF is usually used as secondary treatment of combined biological treatment where it removes carbonaceous substrates and suspended solids, and BANF is based on the immobilization of microorganisms on the media. In the leachate treatment process, Fenton oxidation was applied for COD removal and to enhance biodegradability of refractory organics, while BANF-BAF process was applied to remove total nitrogen (TN), carbonaceous substrates, suspended solids and to further remove COD. The landfill leachate to be used in the pilot study was collected from a municipal landfill in Hangzhou city, Zhejiang Province, China.
4.2.1 Methodology
Figure 4.5 shows the schematic set up of the pilot study. The leachate tank was connected to the Fenton reactor, subsequently to an up flow BANF column and finally to the up flow BAF column. There were two stages of the Fenton-BANF-BAF treatment process, to ensure the effluent would meet the new discharge standards.

4.2.2 Results
Ideal operating parameters of the Fenton process to maximise efficiency of the Fenton reagents were obtained. The indicated maximal COD removal efficiency or about 77.1% with mass ratio H2O2/COD was 1.5:1 and molar ratio Fe2+:H2O2 is 1:1.
After Stage 1 treatment process, the COD of the Fenton effluent was about 308mg/L and after Stage 2 treatment process, the COD of the Fenton effluent was about 109mg/L. Figure 4.6 compares the influent and effluent COD as well as the removal efficiency at each stage.
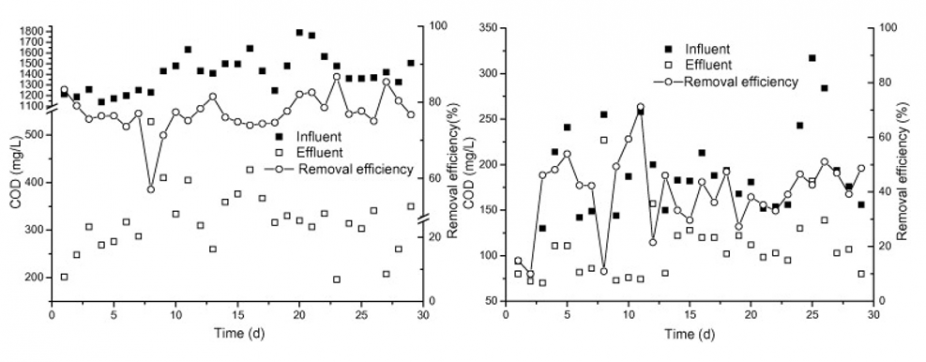
The removal efficiency of TN in the first stage was around 68 +/- 14% depending on the load variation. Figure 4.7 compares the influent and effluent TN as well as the removal efficiency at each stage. As there were potential TN concentration variability, a second stage was necessary to ensure effluent TN to meet discharge standard.

Considering both Stage 1 and 2 in the Fenton BANF-BAF process in treating the leachate, it was found that the COD removal efficiency was 96.4% while the TN removal efficiency was 96.0%. The effluent after completing all treatment stages were well within the new discharge limits. It can be observed that the Fenton process was mostly responsible for COD removal efficiency whereas BANF-BAF process was mostly responsible for TN removal efficiency. The characteristics of effluent and removal efficiency are showed in Figure 4.8 and Table 4.4.
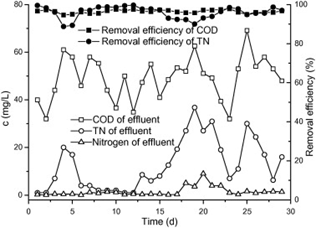
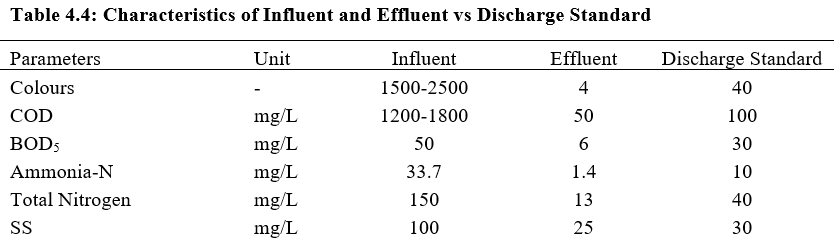
4.2.3 Discussion
Although the contribution of Fenton reaction contributed >90% of the COD removal, the application of BANFs and BAFs was important for meeting the COD, TN, and ammonium discharge standard of China. Results show that this two-stage Fenton-BANF-BAF process has high COD and TN removal capability in treating leachate in landfills and is sufficient in meeting the new guidelines set by the Pollution Control on the Landfill Site of Municipal Solid (GB16889-2008) of China, as well as being able to resist considerable variation in influent characteristics.
4.3 Pilot Study in Tallinn, Estonia
One main drawback of widespread application of the Fenton Process is the formation of large amounts of ferric sludge. A pilot plant study done by Klein et al. (2017) in Estonia was conducted to evaluate a Fenton-like leachate treatment process which has reduced production of ferric sludge while maintaining the efficiency of combined landfill leachate treatment process. The leachate in the landfill used for this study had a high proportion of nitrogen and relatively low concentration of phosphorous. In addition, high lignin and tannins value indicated the presence of substantial amount of bio-recalcitrant COD.
4.3.1 Methodology
A combined three-stage pilot plant was set up for this study as shown in Figure 4.9, which included an activated sludge pre-treatment tank (BIO1), a chemical treatment tank (CHEM) and an activated sludge post-treatment tank (BIO2). Dissolved oxygen concentration, pH, mixing rate, pump rate, aeration and settling period in the tanks were kept constant through automation. Biologically pre-treated supernatant was collected in the first buffet tank, after which it was pumped into the chemical treatment reactor.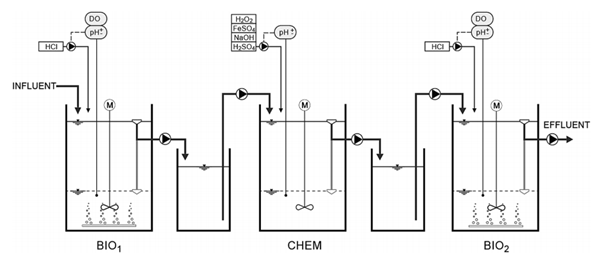
4.3.2 Results
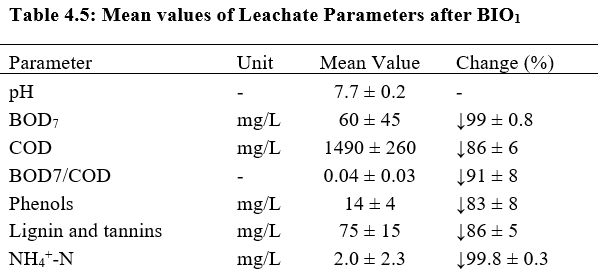
In the present study, to avoid solids accumulation during the ferric sludge reuse cycles and to increase overall efficiency of the combined process, biological pre-treatment (i.e., BIO1) was applied to remove suspended solids, easily biodegradable organics, and ammonium from the wastewater. While the substrate concentrations of the influent varied substantially, the effluent parameters of the first activated sludge reactor were rather stable; BOD7 removal efficiency was 99% and COD removal efficiency 86%. Biological pre-treatment removed 99 % of NH4+–N, 83 % of phenols and 86 % of the lignin and tannins. Table 4.5 illustrates the mean values of various parameters after biological pre-treatment process.
The next stage of treatment is the Fenton process. Precision of chemical dosage will affect the efficiency of the Fenton Process and higher Fenton reagent doses will tend to lead to higher COD removal efficiency. Based on literature research, optimal application of COD/H2O2/Fe2+ by mass is 1/4/0.8, which has most efficient COD removal and biodegradability.

The chemical treatment was efficient in removing recalcitrant organic substances. During the operational period, the BOD7 increased by about 65% on average which is due to recalcitrant substances being degraded into easily biodegradable compounds. The increase in ammonium concentrations can be explained by the release of ammonia from oxidised biomolecules.
Finally, as the effluent of chemical treatment contained substantial concentrations of organics, the wastewater needed an additional treatment stage before discharge. Therefore, the biologically and chemically treated landfill leachate was subjected to a biological post-treatment reactor (BIO2) for final purification. Table 4.7 illustrates the mean values of various parameters after post-treatment process.

4.3.3 Discussion
This study investigated landfill leachate purification using a three-stage process combining the activated sludge pre-treatment. Although the combination of activated sludge treatment and Fenton process proved efficient in leachate purification, substantial amount of ferric sludge was produced in chemical treatment stage. This Fenton-like three-stage process can help reduce waste through addition of ferric catalysts, maintain removal efficiency of undesirable compounds, and can be implemented on an industrial scale.
4.4 Pilot Study in Virginia, United States of America
A pilot study done by Liu, Novak, and He (2020) was conducted to investigate improvements in the Fenton process for leachate treatment to reduce the total nitrogen using a Membrane Electrochemical Reactor (MER). The MER was developed in a separate study (Liu, Novak, & He, 2019). The landfill leachate was obtained from a municipal solid waste landfill in Virginia, USA, with characteristics of substantial dissolved organic matter such as humic acid (HA) and fulvic acid. In this study, apart from Fenton process, precipitation, and recovery of HA was accomplished to reduce concentration of dissolved organic matter, and MER treatment of leachate was done to reduce total nitrogen.
4.4.1 Methodology
The experiments were conducted in three cases variations as shown in Figure 4.10. Case A was direct application of Fenton process for treating raw leachate at pH 3 as a control group for comparison as pH 2-4 is considered as the optimum pH range for traditional Fenton oxidation. In Cases B and C, HA precipitation and recovery were incorporated before Fenton oxidation. In Case B, raw leachate pH was reduced below 2 by acid addition to separate HA from the leachate by precipitation, followed by Fenton process at pH 3. In Case C, MER was added to the treatment system, followed by HA precipitation (pH 2) and Fenton oxidation at pH 3. After the Fenton process, the solution pH was adjusted to 7 for coagulation and sludge separation.

During the Fenton Process, the leachate (raw leachate for Case A and pre-treated leachate for Case B and C) was acidified to a pH of 3. The reagent molar ratio was fixed at Fe2+: H2O2 = 1: 3, which optimises oxidation of leachate with the maximized treatment efficiency and minimized Fe2+ consumption and iron sludge production.
4.4.2 Results
In the raw leachate, 86.2 ± 7.2% of the initial total nitrogen concentration was ammonia nitrogen. In Case C, the TN removal was 89.1 ± 2.9% after 4-h MER treatment while the control reactor had only 38.0 ± 4.3% removal. The MER treatment decreased the ammonium concentration in the leachate (anolyte) from 2500 ± 200 mg/L to 180 ± 70 mg/L, representing a removal efficiency of 92.6 ± 3.1%.
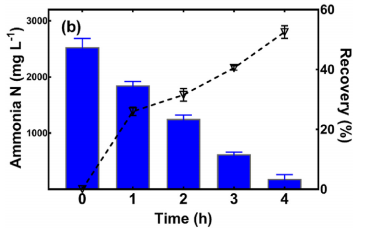
Regarding HA Recovery, Figure 4.11 shows that 1.38 ± 0.26 g of HA (per 1 L of raw leachate) was recovered in Case B, higher than 0.56 ± 0.12 g in Case C. This difference is attributed to the HA degradation by MER treatment in Case C.
Regarding COD Removal, Case C had a larger COD removal increase (22.1%) with increasing reagents dose than that for Case A (13.8%) and Case B (17.5%), indicating the more effective MER-Fenton synergy at a high Fenton reagents dose. Figure 4.12 shows that COD removal efficiency was greatest for Case C and lowest for Case A.
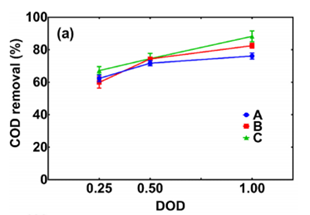
Compared to Case A that had direct Fenton treatment of raw leachate, Case C with a two-stage system of MER-Fenton treatment had 21% greater COD removal. The higher COD removal in Case B than that of Case A suggested that HA removal before the Fenton process can allow better oxidation of other organics, possibly because humic acids have a high electron-donating capacity to consume hydroxyl radicals from Fenton oxidation (Aeschbacher, Graf, Schwarzenbach, & Sander, 2012) and thus the reduction of HA would allow more radicals available for oxidizing other organic matter during Fenton oxidation.
As expected, Fenton process exhibited limited ammonia removal from the raw leachate (Case A) with removal efficiency varied from 13 ± 2.8% to 21.5 ± 3.6%. Incorporating HA recovery in Case B significantly increased ammonia removal efficiency to 54.5 ± 2.8%. The addition of the MER in Case C achieved the highest ammonia removal efficiency of 97.5 ± 3.7%. These results demonstrate that the joint efforts between the MER and HA recovery could greatly assist with the Fenton process to accomplish ammonia removal, which cannot be done by the Fenton process alone.
4.4.3 Discussion
The synergistic effects between MER and Fenton process have increased leachate COD and ammonia removal to 88.3% and 97.5%, compared to the standalone Fenton process. In this pilot study, the coupled system also greatly lowered the chemical dose requirement as well as the overall cost, mainly due to the decreased organics in the leachate and elimination of acid requirement.
Chapter 5: Conclusions from Pilot Studies
The Fenton process, as exemplified unanimously across all pilot studies, is an integral component in landfill leachate control and treatment, due to its high COD removal efficiency and cost-effectiveness. While leachate composition in landfills around the world varies due to climate and economic conditions, the input reagent ratio for the Fenton Process can be adjusted to maximise treatment efficacy for any condition on site.
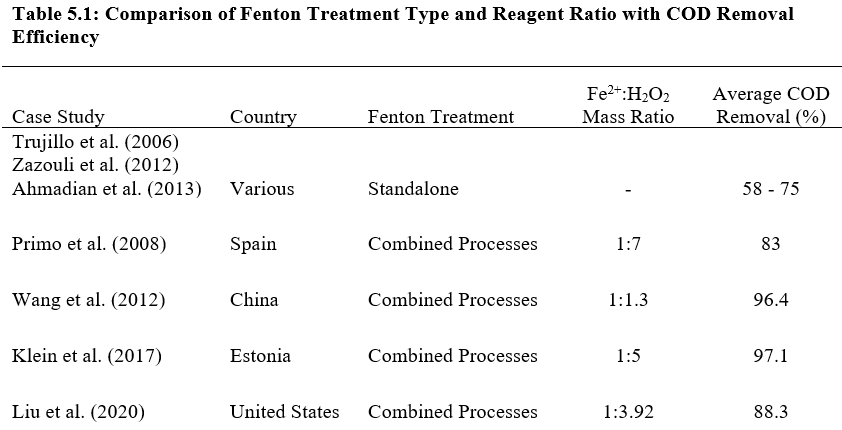
A range of Fe2+/H2O2 Fenton reagent mass ratio was used across all pilot studies to optimise the Fenton process due to different conditions on-site. Table 5.1 shows the different Fenton reagent mass ratios used in the different case studies, the corresponding COD removal, as well as the contrast in efficiency obtained from standalone Fenton treatment through literature review. Combined treatment systems with the Fenton Process show greater COD removal efficiency than standalone Fenton process which can be attributed to synergistic effects between the different treatment processes.
To maintain robust leachate treatment efficacy, however, requires additional processes along with the Fenton process. The pilot studies from Spain, China, Estonia, and United States illustrate that combined treatment systems with the Fenton process can effectively work well in tandem to increase the efficacy of leachate treatment as well as to reduce waste. The findings are described as follows:
- The Fenton process is capable of substantially removing COD in leachate, but this process alone cannot remove various compounds such as ammonium and therefore not sufficient in passing stringent discharge regulations in many territories.
- A secondary standalone Fenton Treatment step does not have a significant impact on COD removal as the remaining organic material after the primary Fenton oxidation step will be incalcitrant to secondary oxidation.
- Additional indirect treatment processes such as the reduction of humic acid in leachate may enhance the efficacy of the Fenton Process.
- The Fenton process generates substantial amount of ferric sludge; however, it is possible to reduce ferric waste generation through catalyst reaction and be implemented on an industrial scale.
At its core, the Fenton process has worked very well in leachate control and treatment in landfill sites around the world. The Fenton process provides substantial treatment in COD removal and can easily be adapted into any landfill sites through adjustments and optimisation in the Fenton reagent ratio. The pilot plant studies described in this report show that there are many other additional treatment processes that can complement and enhance the efficacy of the Fenton process with largely successful results and can be considered for large-scale implementation on landfills.
References
Aeschbacher, M., Graf, C., Schwarzenbach, R. P., & Sander, M. (2012). Antioxidant properties of humic substances. Environmental science & technology, 46(9), 4916-4925.
Ahmadian, M., Reshadat, S., Yousefi, N., Mirhossieni, S. H., Zare, M. R., Ghasemi, S. R., . . . Fatehizadeh, A. (2013). Municipal leachate treatment by Fenton process: effect of some variable and kinetics. Journal of environmental and public health, 2013.
Alfaya, E., Iglesias, O., Pazos, M., & Sanromán, M. A. (2015). Environmental application of an industrial waste as catalyst for the electro-Fenton-like treatment of organic pollutants. RSC Advances, 5(19), 14416-14424.
Babuponnusami, A., & Muthukumar, K. (2014). A review on Fenton and improvements to the Fenton process for wastewater treatment. Journal of Environmental Chemical Engineering, 2(1), 557-572.
Byrne, W. (n.d.). What You Need To Know About Landfill Leachate Treatment. OXYMEM.
Costa, A. M., Alfaia, R. G. d. S. M., & Campos, J. C. (2019). Landfill leachate treatment in Brazil–An overview. Journal of environmental management, 232, 110-116.
Domingues, E., Gomes, J., Quina, M. J., Quinta-Ferreira, R. M., & Martins, R. C. (2018). Detoxification of olive mill wastewaters by Fenton’s process. Catalysts, 8(12), 662.
Jung, Y. S., Lim, W. T., Park, J. Y., & Kim, Y. H. (2009). Effect of pH on Fenton and Fenton‐like oxidation. Environmental technology, 30(2), 183-190.
Klein, K., Kivi, A., Dulova, N., Zekker, I., Mölder, E., Tenno, T., . . . Tenno, T. (2017). A pilot study of three-stage biological–chemical treatment of landfill leachate applying continuous ferric sludge reuse in Fenton-like process. Clean Technologies and Environmental Policy, 19(2), 541-551.
Liu, X., Novak, J. T., & He, Z. (2019). Removal of landfill leachate ultraviolet quenching substances by electricity induced humic acid precipitation and electrooxidation in a membrane electrochemical reactor. Science of The Total Environment, 689, 571-579.
Liu, X., Novak, J. T., & He, Z. (2020). Synergistically coupling membrane electrochemical reactor with Fenton process to enhance landfill leachate treatment. Chemosphere, 247, 125954.
Pouran, S. R., Aziz, A. A., & Daud, W. M. A. W. (2015). Review on the main advances in photo-Fenton oxidation system for recalcitrant wastewaters. Journal of Industrial and Engineering Chemistry, 21, 53-69.
Primo, O., Rueda, A., Rivero, M. J., & Ortiz, I. (2008). An integrated process, Fenton reaction− ultrafiltration, for the treatment of landfill leachate: pilot plant operation and analysis. Industrial & engineering chemistry research, 47(3), 946-952.
Qasim, S. R., & Chiang, W. (1994). Sanitary landfill leachate: generation, control and treatment.
Ranjit, P. J. D., Palanivelu, K., & Lee, C.-S. (2008). Degradation of 2, 4-dichlorophenol in aqueous solution by sono-Fenton method. Korean Journal of Chemical Engineering, 25(1), 112-117.
Renou, S., Givaudan, J., Poulain, S., Dirassouyan, F., & Moulin, P. (2008). Landfill leachate treatment: Review and opportunity. Journal of hazardous materials, 150(3), 468-493.
Stegmann, R., Heyer, K., & Cossu, R. (2005). Leachate treatment. Paper presented at the Proceedings Sardinia.
Trujillo, D., Font, X., & Sánchez, A. (2006). Use of Fenton reaction for the treatment of leachate from composting of different wastes. Journal of hazardous materials, 138(1), 201-204.
Vergara, S. E., & Tchobanoglous, G. (2012). Municipal solid waste and the environment: a global perspective. Annual Review of Environment and Resources, 37, 277-309.
Wang, X., Han, J., Chen, Z., Jian, L., Gu, X., & Lin, C.-J. (2012). Combined processes of two-stage Fenton-biological anaerobic filter–biological aerated filter for advanced treatment of landfill leachate. Waste Management, 32(12), 2401-2405.
Xu, M., Wu, C., & Zhou, Y. (2020). Advancements in the Fenton process for wastewater treatment. Advanced Oxidation Processes, 61.
Yidong, G., Xin, C., Shuai, Z., & Ancheng, L. (2012). Performance of multi-soil-layering system (MSL) treating leachate from rural unsanitary landfills. Science of The Total Environment, 420, 183-190.
Zazouli, M. A., Yousefi, Z., Eslami, A., & Ardebilian, M. B. (2012). Municipal solid waste landfill leachate treatment by fenton, photo-fenton and fenton-like processes: Effect of some variables. Iranian journal of environmental health science & engineering, 9(1), 1-9.





























3 COMMENTS
Xiaolan Zeng*
May, 07, 2021 This is a good work on introduction of Fenton process and its application to treat landfill leachate. There are several suggestions to improve it for more scientific and objective. 1. For leacheat, it could come from the degradation of MSW itself,especially organic materials, besides "the percolation of water outside the landfill".2.Before talking the possible contamination routes of leachate, it is necessary to summarize how the leachate is dangerous to our environment and human health.3. It is better to indicate that the special water quality of leachate results in failing to meet the discharge standard with classic wastewater treatment processes by comparing to the quality of domestic wastewater. 4.Pay more attention to the terms " landfill" and "sanitary landfill" which may not the same conceptions. The former can include simple landfill, controlled landfill, sanitary landfill,etc..5. AOPs including Fenton process are usually applied to be the advanced treatment process of leachate to reduce the refractory organics followed biological treatment process,and with the result of BOD5/COD ratio increasing. It will be helpful to understand Fenton process for leachate treatment if the roles of AOPs and biological process in wastewater treatment are clearly stated.6. For the first pilot study(in Cantabria, Spain), at the beginning, it was introduced that the study was to "evaluate the viability of Fenton treatment as pre-treatment to increase the biodegadability of leachate", but no words were related to this point in the following content.7. The review work will be more logical if the contribution for Fenton process of each pilot study performed in Spain,China,Estonia and the United States of America could be emphasized and summarized.Xiaolan Zeng*
May, 07, 2021 This is a nice work! There are some suggestions in my last comments and hope you can receive them. Good luck!Alexandra B. Ribeiro
Jan, 27, 2023 Congratulations! This is a nice work.I suggest you summarize the 4 pilot studies.LEAVE A COMMENT