Landfill Leachate Treatment
Contents [show]
Introduction
The sanitary landfill method for disposal of solid waste is widely used due to its economic advantages in terms of exploitation and capital costs (Renou et al., 2008). In addition to the economic advantages, landfills enable controlled decomposition of waste to final stabilized material (Renou et al., 2008). However, there are concerns related to leachate produced from landfills, which can migrate to and contaminate the surface and groundwater and soil environment.
Landfill leachate contains various contaminants (i.e. organic and inorganic pollutants), of which the composition and content varies with geographic location, waste composition, age, pH, moisture content, and other landfill site characteristics (Bove et al., 2015). Leachate requires pretreatment on-site to meet standards to discharge to wastewater treatment plants (WWTPs) or directly into surface water (Abbas et al., 2009). In any case, available treatment technologies must be carefully reviewed to select the most feasible and efficient process.
In this report, factors that affect characteristics of leachate are described to identify the difference in composition according to variables such as waste/leachate age. Three main categories of leachate treatment technologies will be reviewed (i.e. leachate transfer, biological and physicochemical methods), with a focus on the suitability of each technology on different types of leachate. Finally, a rising issue regarding contaminants of emerging concern in leachate is discussed to address future implications of closed and ongoing landfill sites.
Landfill Leachate Generation and Composition
Leachate is generated in the landfill depending primarily on moisture content and field capacity (Equations 1-2) (Zornberg et al., 1999), which refers to the maximum attainable moisture content without downward percolation (El-Fadel et al., 2002). The moisture-holding capacity of a given landfill is influenced by the field capacity, wilting point, and porosity (Figure 1). When moisture content exceeds field capacity due to precipitation, decrease of voids, etc., leachate is formed as the gravitational force overcomes the retention force resulting in vertical flow of liquids through the landfill (Equation 3).
θw = Vwater / Vwaste (Equation 1)
θw: moisture content
Vwater: volume of water
Vwaste: volume of waste
θFC = 21.7 * lnγt - 5.4 (Equation 2)
θFC: field capacity (%)
γt: total (wet) unit weight of waste (kN/m3)

Figure 1. Volumetric Phase Relationships and Moisture Retention Parameters in Porous Material (Zornberg et al., 1999)
The amount of leachate generated in the landfill depends mainly on water availability (e.g. precipitation, surface water, leachate recirculation), characteristics of final cover (soil and vegetation type, impermeable cover, slope), and characteristics of waste (moisture content, density) (Canziani and Cossu, 1989). If the landfill site properties favor infiltration, more leachate will be generated (Figure 2). Other properties of the landfill and surrounding site can also affect leachate formation (Table 1).

Figure 2. Schematic of the general hydrological balance in a completed sanitary landfill with leachate drainage system. P: precipitation; J: irrigation or leachate recirculation; R: surface runoff; R*: runoff from external areas; ET: evapotranspiration; Pi=P+J+R*–R–ET±∆US; US: water content in soil; UW: water content in waste; S: water added by sludge disposal; b: water production (if>0) or consumption (ifS, IG: water from natural aquifers; Pi=S+IS+IG; L=Pi±∆Uw+b: total leachate production; Li: infiltration into aquifers; LR: leachate collected by drains (Canziani and Cossu, 1989)
The composition and physicochemical properties of landfill leachate is affected by a number of factors (Table 1), but is mainly determined by the landfilled waste and biodegradation phase, which is dependent on landfill age (El-Fadel et al., 2002; Schiopu and Gavrilescu, 2010).
Table 1. Factors influencing leachate formation and composition in landfills (El-Fadel et al., 2002)
| Climatic & Hydrogeologic | Rainfall, snowmelt, groundwater intrusion |
| Site Operations & Management | Refuse pretreatment, compaction Vegetation, cover, sidewalls & liner material Irrigation, recirculation, liquid waste co-disposal |
| Refuse Characteristics | Permeability, age Particle size, density Initial moisture content Composition |
| Internal processes | Refuse settlement Organic material decomposition Hydrolysis, adsorption, biodegradation Speciation, dissolution, dilution Ion exchange, redox Contact time, partitioning, precipitation Gas & heat generation & transport |
There are five distinct phases of biodegradation observed in landfills, which are characterized by major biochemical reactions and their products (Figure 3) (Christensen and Kjeldsen, 1989; Schiopu and Gavrilescu, 2010):
(Phase I) Aerobic phase: readily biodegradable organic matter is aerobically decomposed (H2O, CO2)
(Phase II) Acidogenic phase: hydrolysis, fermentation and activity of acetogenic bacteria is predominant (CO2, H2, NH4+, organic acids)
(Phase III) Acetogenic phase: methanogenic bacteria start to grow slowly (acetic acid, CO2, H2)
(Phase IV) Methanogenic phase: methane is produced at a stable rate (landfill gas: 60% CH4, 40% CO2)
(Phase V) Aerobic phase: methane production rate decreases and nitrogen appears due to diffusion from the atmosphere (H2O, CO2)

Figure 3. Developments in gas and leachate composition in a landfill cell (Christensen and Kjeldsen, 1989)
Each biodegradation stage can be identified by examining the concentration of different indicators (i.e. chemical oxygen demand (COD), ammonia nitrogen (NH4+-N), dissolved solid (DS), suspended solid (SS), xenobiotic organic compounds (XOCs), heavy metals, salts) (Schiopu and Gavrilescu, 2010). These indicators may be similar to those found in conventional municipal wastewater. However, landfill leachate generally has a comparatively diverse mixture of contaminants at higher concentrations (Schiopu and Gavrilescu, 2010), and requires a different or modified treatment method than those used for municipal wastewater.
Leachate formed from municipal solid waste is composed of various organic and inorganic contaminants (table 2). Its composition may vary depending on the phase of biodegradation, which also is affected partially by landfill age as mentioned (Table 3).
Table 2. Chemical composition of leachate from municipal solid waste (El-Fadel et al., 2002)
| Parameter | Concentration range (mg/L) | Parameter | Concentration range (mg/L) |
| Alkalinity (as CaCO3) | 0–20,850 | Nitrogen (ammonia) | 0–1250 |
| Aluminum | 0.5–85.0 | Nitrogen (Kjeldahl) | 0–3,320 |
| Antimony | 0–3.19 | Nitrogen (nitrate) | 0–9.8 |
| Arsenic | 0–70.2 | Nitrogen (nitrite) | 0–1.46 |
| Barium | 0–12.5 | Nitrogen (organic) | 0–1,000 |
| Benzene | 0.1–0.6 | Organic halides | 0.320–3.5 |
| Beryllium | 0–0.36 | pH (unit) | 1.5–9.5 |
| BOD5a | 480–72,500 | Phenol | 0.17–6.6 |
| Boron | 0.413 | Phenols | 0–4 |
| Cadmium | 0–1.16 | Phosphate | 0.01–154 |
| Calcium | 5–4,080 | Phosphorus (total) | 0–234 |
| Chloride | 11,375 | Potassium | 0.16–3,370 |
| Chromium | 0–22.5 | Selenium | 0–1.85 |
| CODb | 0–195,000 | Silver | 0–1.96 |
| Conductivity (mmho/cm) | 480–72,500 | Sodium | 0–8,000 |
| Copper | 0–9.9 | Sulfate | 0–1,850 |
| Cyanide | 0–6 | TDSc | 584–55,000 |
| Ethylbenzene | 0–4.9 | Thallium | 0–0.32 |
| Fluoride | 0.1–1.3 | Tin | 0–0.16 |
| Hardness (as CaCO3) | 0.1–225,000 | TOCd | 335,000 |
| Iron | 0–42,000 | Toluene | 0–3.2 |
| Lead | 0–14.2 | TSSe | 140,900 |
| Magnesium | 0–115,600 | Turbidity | 40–500 |
| Manganese | 0.05–1,400 | TVAf (as acetic acid) | 0–19,000 |
| Mercury | 0–3 | Zinc | 0–1,000 |
| Nickel | 0–7.5 |
aBiochemical oxygen demand
bChemical oxygen demand
cTotal dissolved solid
dTotal organic carbon
eTotal suspended solid
fTotal volatile acid
Table 3. Composition of leachate depending on age of landfill (Bove et al., 2015)
| Country | COD (g/m3) | BOD (g/m3) | BOD/COD | pH | TN (g/m3) | NH4+-N (g/m3) |
| Young landfills | ||||||
| Canada | 13,800 | 9,660 | 0.70 | 5.8 | 212 | 42 |
| China | 15,700 | 4,200 | 0.27 | 7.7 | - | 2,260 |
| 8,528 | 5,669 | 0.66 | 8.0 | 1,384 | 1,154 | |
| Greece | 70,900 | 26,800 | 0.38 | 6.2 | 3,400 | 3,100 |
| Italy | 19,900 | 4,000 | 0.20 | 8.0 | - | 3.917 |
| Turkey | 20,000 | 11,000 | 0.55 | 7.8 | - | 2,500 |
| Averagea | 24,805±22,982 | 10,221±8,619 | 0.46±0.21 | 7.2±1.0 | 1,665±1,612 | 2,162±1,385 |
| Medium-age landfills | ||||||
| China | 5,800 | 430 | 0.07 | 7.6 | - | - |
| Germany | 3,180 | 1,060 | 0.33 | - | 1,135 | 840 |
| Greece | 5,350 | 1,050 | 0.20 | 7.9 | 1,100 | 940 |
| Hong Kong | 6,610 | 1,600 | 0.24 | 6.4 | 2,000 | 1,500 |
| Italy | 5,050 | 1,270 | 0.25 | 8.4 | - | 1,130 |
| Poland | 1,180 | 331 | 0.28 | 8.0 | - | 743 |
| Turkey | 9,500 | - | - | 8.1 | 1,450 | 1,270 |
| Averagea | 5,239±2,618 | 957±490 | 0.23±0.09 | 7.7±0.7 | 1,421±416 | 1,070±285 |
| Old landfills | ||||||
| Brazil | 3,460 | 150 | 0.04 | 8.2 | - | 800 |
| 5,414 | 813 | 0.15 | 8.2 | 2,036 | 1,811 | |
| China | 1,703 | - | - | 8.5 | 2,117 | 1,972 |
| 1,819 | - | - | 8.8 | 1,055 | 1,040 | |
| 2,200 | 100 | 0.05 | 8.0 | 2,030 | 2,000 | |
| Finland | 556 | 62 | 0.11 | - | 192 | 159 |
| France | 500 | 7.1 | 0.01 | 7.7 | 540 | 430 |
| Germany | 1,225 | 290 | 0.26 | - | - | 445 |
| Greece | 2,456 | - | - | 8.4 | 357 | 238 |
| Italy | 1,759 | 301 | 0.17 | 8.0 | 1,191 | 1,061 |
| Poland | 757 | 132 | 0.17 | - | 397 | 362 |
| 732 | 51 | 0.07 | 8.1 | 420 | 340 | |
| Spain | 4,512 | 558 | 0.12 | - | 2,199 | 1,623 |
| 4,357 | 810 | 0.19 | 8.1 | 4,058 | 3,772 | |
| 3,921 | 123 | 0.03 | 8.5 | 6,317 | 5,975 | |
| 6,200 | 832 | 0.13 | 8.3 | 2,305 | 2,003 | |
| Vietnam | 3,621 | 425 | 0.12 | 8.4 | 3,868 | 3,449 |
| Averagea | 2,652±1,786 | 332±304 | 0.12±0.07 | 8.2±0.3 | 1,939±1,715 | 1,616±1,557 |
aAverage values calculated based on those reported in the literature ± standard deviation.
Leachate Treatment
Factors to Consider
Leachate generated in landfill is collected and treated by one or more of the following strategies depending on various parameters (Schiopu and Gavrilescu, 2010; Townsend et al., 2015):
| On-site treatment and discharge | Treatment system is constructed on-site or leachate is recycled on-site |
| On-site pretreatment followed by off-site treatment | Some off-site facilities may have limits that require pretreatment |
| Transport and treatment off-site | Leachate is transported via tanker truck or pumped directly through a pipe to off-site wastewater treatment facility |
The following factors are taken into account when applying technologies to treat leachate (Abbas et al., 2009; Schiopu and Gavrilescu, 2010; Townsend et al., 2015):
- Leachate composition, properties, and volume
- Need for leachate storage
- Local water standards requirement for discharge
- Technical value, ease of implementation, and cost effectiveness of technologies
Treatment Technology
Leachate Transfer
The three main categories of conventional landfill leachate treatment technologies are (1) leachate transfer, (2) biological, and (3) physicochemical methods (Renou et al., 2008).
Co-Treatment with Municipal Sewage
Leachate collected at the landfill can be treated by combining with municipal sewage at the sewage treatment plant. This method is used for its easy and low-cost operation (Renou et al., 2008). However, leachate co-treatment has limitations due to high contaminant load, organics with low biodegradability, and presence of heavy metals (Quasim and Chiang, 1994; Booth et al., 1996, Çeçen and Aktaş, 2004), which may lead to reduction in the performance of the sewage treatment plant. In addition, concerns were raised regarding the effect of simple dilution rather than treatment of the leachate.
Leachate Recirculation
Leachate can be recycled by recirculating it through the landfill to improve biodegradation and waste stabilization by controlling the moisture content (Renou et al., 2008). The bioreactor landfill is an enhanced system with controlled leachate collection and injection, which is often supplemented with other liquids to maintain moisture content near field capacity to optimize decomposition (Figure 4) (EPA, 2017). Advantages of leachate recirculation or bioreactor landfill are increased decomposition rate, lower leachate treatment/disposal costs, and shortened post-closure maintenance period. Disadvantages include increased gas emissions and odors, physical instability of waste mass, and surface seeps. (EPA, 2017).
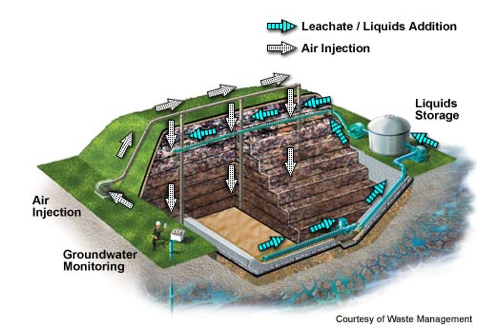

Figure 4. Schematic of aerobic (left) and anaerobic (right) bioreactor landfills (EPA, 2017)
Constructed Wetlands
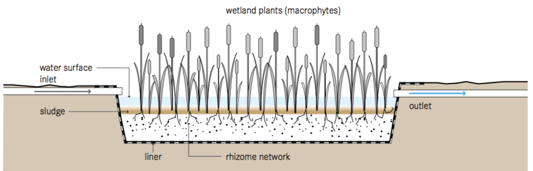
Another on-site treatment technique is constructed wetland, where leachate is treated by phytodepuration or reused as a fertilizer for energy crops (Justin and Zupančič, 2009; Bove et al., 2015). There are three types of systems, free-flow systems, horizontal subsurface flow systems, and vertical subsurface flow systems (Figure 5).
(a)
(b)

(c)

Figure 5. Schematic of constructed wetland systems: (a) free-flow system, (b) horizontal subsurface flow system, and (c) vertical subsurface flow system (Tilley et al., 2014)
The advantages of this method are low construction and operational costs, low power consumption, and high contaminant removal efficiency (Bove et al., 2015). The disadvantages are space requirement, generation of aerosols, applicability on low volumes only, and difficulty of control (Bove et al., 2015; Schiopu and Gavrilescu, 2010).
Biological Processes
There are many types of biological processes used for leachate treatment, and most of them are applied for wastewater treatment processes. Biological processes are generally classified by aeration (aerobic, anaerobic, or combined), media (activated sludge, biofilm, or hybrid) or reactor type (continuous flow stirred-tank reactor, sequencing batch reactor, or ditch) (Metcalf & Eddy / Aecom, 2014). Biological treatment is used for its reliability, simplicity, high cost-effectiveness, and its efficiency in removing organic compounds.
Aerobic treatment processes
Aerobic treatment processes should allow to treat biodegradable organics and nutrients. Unlike anaerobic processes, nitrification (oxidizing ammonium to nitrate) is available in the aerobic processes. The aerobic processes that are based on suspended growth (i.e., activated sludge) have been widely studied and adopted (Lin and Chang, 2000). The biological processes with attached growth (biofilm) have recently attracted significant interest: biological aerated filter (BAF), moving-bed biofilm reactor (MBBR) (Renou et al., 2008).
The integration of aerobic processes and membrane separation technology which is termed membrane bioreactor (MBR) has also led to new focus on treatment of leachate (Renou et al., 2008). The biological aerobic treatment processes are as follow:
Suspended growth processes: Aerated lagoon, activated sludge process, SBR, MBR, etc..
Attached growth processes: Trickling filter, BAF, MBBR, FBBR, etc..

Figure 6. Schematic setup of an aerobic moving-bed biofilm reactor (MBBR) with suspended cells (Bove et al., 2015)
Anaerobic treatment processes
Anaerobic treatment processes are commonly applied (e.g. digestion), and these processes are suitable for dealing with high strength wastewater (Renou et al., 2008; Prokhel and Viraraghavan, 2004). Anaerobic digestion conserves energy and produces less sludge compared to aerobic processes with suspended growth. However, anaerobic digestion suffers from low reaction rates (Berrueta et al., 1992). In addition to anaerobic digestion, there are other processes such as up-flow anaerobic sludge blanket (UASB) reactor and anaerobic filter (AF). Oxidized ammonium and nitrate, can be denitrified to nitrogen gas in anaerobic condition, therefore, aerobic processes and anaerobic processes can be combined for completely treating nitrogen as nutrients.

Figure 7. A small-scale anaerobic digester (Agri-Food and Biosciences Institute, 2016)
Physicochemical Processes
Physicochemical treatment processes are more efficient ways to solve landfill leachate problems than biologic treatment against certain substances. Various kinds of physicochemical processes have their own advantages in leachate treatment (Table 5). (Torretta et al., 2017)
Flocculation-Coagulation
Flocculation is a treatment typically applied to remove non-settleable colloidal solids (such as surfactants, heavy metals, fatty acids and humic acids) using flocculants such as FeCl3 (Torretta et al., 2017), which is more suitable for old and stable leachates (Renou et al., 2008). Flocculation is often used upstream of biological treatment or reverse osmosis, or downstream for the purpose of final cleaning. The main coagulants are aluminum sulfate, ferrous sulfate, ferric chloride and chlorine ferric sulfate (Amokrane et al., 1997). Flocculation is suitable for landfill leachate treatment due to its strength against low pH and high organics concentration in the leachate (Torretta et al., 2017). However, this treatment presents some disadvantages: consistent sludge volume is produced and an increase in the concentration of aluminum or iron in the liquid phase may be observed (Silva et al., 2004).
Separation through Membrane Filtration
Membrane separation process is a physical process that can be classified into micro-filtration (MF), ultra-filtration (UF), and nano-filtration (NF) based on the size of the membrane pores (Figure 8) (Torretta et al., 2017). Reverse osmosis (RO) is another filtration technology that is driven by inverse pressure across a semi-permeable membrane commonly applied for water desalination or drinking water plants (Torretta et al., 2017). Membrane separation treatments, especially NF and RO, are very efficient in decreasing the COD, SS, and organics in landfill leachate (Torretta et al., 2017). The disadvantages of membrane filtration are high pressure requirement leading to high energy consumption and membrane fouling which requires surface cleaning processes (Torretta et al., 2017).

Figure 8. Magnitude of Membrane Filtration (KYDEP, 2012)
Air Stripping
Air stripping is often applied in concert with other techniques for ammonia removal (Torretta et al., 2017). In this process, organic constituents are separated from the aqueous phase to the gas phase through exposure to air or steam (Torretta et al., 2017), usually in a stripping tower (Figure 9) (Renou et al., 2008). This technology can remove up to 99.5% of ammonia from contaminated water (Silva et al., 2004), and is excellent for the removal of high levels of ammonia. However, air stripping technology has inherent disadvantages such as possible release of NH3 into the atmosphere and high pH requirement (Renou et al., 2008).
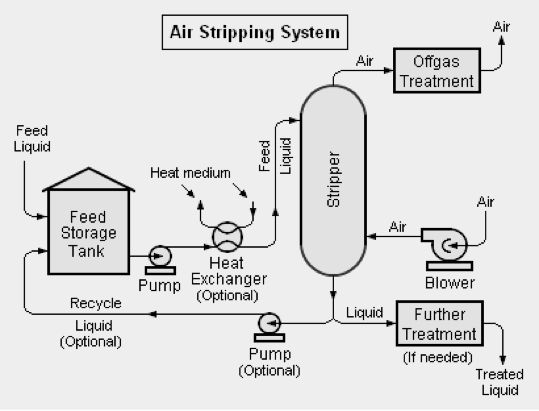
Figure 9. Air stripping system (Beychok, 2010)
Adsorption by Activated Carbon
Adsorption is a surface phenomenon where a fluid (gas or liquid) mixture is attracted and attached to the surface of a solid adsorbent through physical and/or chemical bonds (Foo and Hameed, 2009). Activated carbon (AC) is widely used as an adsorbent primarily due to its large surface area, controllable pore structure, thermostability, pH stability, and the ability to remove both organic and inorganic contaminants from gas or liquid (Figure 10) (Foo and Hameed, 2009). The main disadvantage of the adsorption process is the frequent regeneration of AC columns and consequent AC consumption and cost (Torretta et al., 2017).

Figure 10. Activated carbon column used for leachate treatment (Foo and Hameed, 2009)
Chemical Precipitation
Chemical precipitation is a process in which hydroxide or sulfide salts are used to precipitate target contaminants, i.e. heavy metals and inorganic compounds (Torretta et al., 2017), and is widely used as pre-treatment to remove ammonium nitrogen (NH4+-N) (Renou et al., 2008). This method is suitable for the removal of heavy metals from aqueous solutions due to its simplicity and low cost (Torretta et al., 2017). However, it can also produce a large amount of sludge, which increases treatment of secondary waste (Torretta et al., 2017).
Ion Exchange
Ions and dissolved organic matter from landfill leachate can be removed by ion exchange, where ions are reversibly interchanged between the solid and liquid phases (Figure 11) (Torretta et al., 2017). Synthetic resins with active functional groups, natural zeolites and silicate minerals are widely used to remove heavy metals from aqueous solutions (Torretta et al., 2017). Ion exchange is an environmentally friendly technology with the advantage of possible metal recovery from wastewater, but has high operational cost and require pre-treatment to remove SS (Torretta et al., 2017).
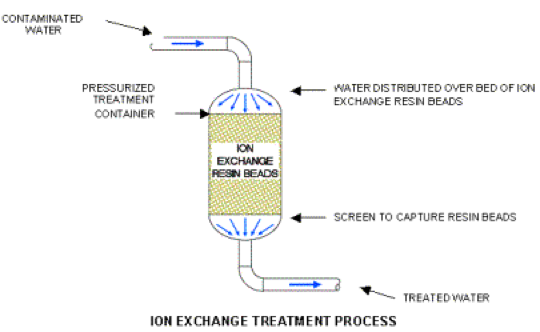
Figure 11. Ion exchange treatment (J&T Technologies, Inc., 2014)
Chemical Oxidation and Advanced Oxidation Processes
Contaminants are mineralized to carbon dioxide, water and inorganics or other harmless products through chemical oxidation (Andreozzi et al., 1999). The main oxidants used for leachate treatment are chlorine, ozone, potassium permanganate and calcium hydrochloride (Abbas et al., 2009; Torretta et al., 2017). Recently, advanced oxidation processes (AOPs), such as Fenton process and photocatalysis (Figure 12), have been suggested as an alternative to conventional chemical oxidation processes (Renou et al., 2008; Torretta et al., 2017). Generation of hydroxyl radicals are increased in AOPs to improve chemical oxidation by using a combination of strong oxidants (e.g. O3, H2O2), irradiation (e.g. ultraviolet, ultrasound, electron beam) and catalysis (e.g. transition metal ions, photocatalyst) (Abbas et al., 2009). AOPs can help oxidize halogenated organics and increase the biodegradability of persistent organic contaminants (Renou et al., 2008). However, they require expensive reactants and thus pre-treatment to reduce COD and control system to monitor treatment performance (Andreozzi et al., 2009; Torretta et al., 2017).

Figure 12. Mechanism of TiO2 photocatalytic oxidation (Hassan et al., 2016)
Comparison of Treatment Processes
Different parameters are needed when selecting leachate treatment methods. One of the factors that influence the application of treatment method is the age of landfill. Table 5 shows the effectiveness of leachate treatment technologies in three kinds of landfill ages: young, medium and old. Leachate transfer and biological processes are effective during the young period of landfills, while the efficiency of different physicochemical processes varies.
Table 5. Effectiveness of leachate treatment depending on landfill age (Abbas et al., 2009)
| Treatment Type | Landfill Age | Target of Removal | Remark | ||
| Young | Medium | Old | |||
| Leachate Transfer | |||||
| Co-Treatment | Good | Fair | Poor | Suspended solid | Excess biomass and nutrients |
| Recycling | Good | Fair | Poor | Improve leachate quality | Least expensive and low efficiency |
| Biological Processes | |||||
| Aerobic Processes | Good | Fair | Poor | Suspended solid | Hamper by refractory compound and excess biomass |
| Anaerobic Processes | Good | Fair | Poor | Suspended solid | Hamper by refractory compound, long time and biogas |
| Physicochemical Processes | |||||
| Coagulation / Flocculation | Poor | Fair | Fair | Heavy metals, suspended solids | High sludge production and subsequent disposal |
| Chemical Precipitation | Poor | Fair | Poor | Heavy metals, NH3-N | Requires further disposal due to sludge generation |
| Adsorption | Poor | Fair | Good | Organic compounds | Carbon fouling can be a problem and granulated activated carbon adsorption is costly |
| Oxidation | Poor | Fair | Fair | Organic compounds | Residual O3 |
| Stripping | Poor | Fair | Fair | NH3-N | Requires other equipment for air pollution control |
| Ion Exchange | Good | Good | Good | Dissolved compounds, cations/anions | Used as a polishing step after biological treatment and treatment cost is high |
| Membrane Filtration | |||||
| Microfiltration | Poor | - | - | Suspended solids | Used after metal precipitation |
| Ultrafiltration | Poor | - | - | High molecular weight compounds | Costly and limited applicability due to membrane fouling |
| Nanofiltration | Good | Good | Good | Sulfate salts, hardness ions | Costly and requires lower pressure than reverse osmosis |
| Reverse Osmosis | Good | Good | Good | Organic and inorganic compounds | Costly and extensive pre-treatment is required prior to RO3. |
Contaminants of Emerging Concern in Leachate: The USGS Findings
Recently, the United States Geological Survey (USGS) conducted a nationwide investigation of contaminants of emerging concern (CECs) in fresh and final leachate generated from 19 and 22 landfill sites, respectively (Masoner et al., 2014; Masoner et al., 2015), in order to assess leachate as a source of CECs in the environment (Figure 14). The details of the research were published in 2014 and 2015 by Masoner et al., and are summarized in the discussion that follows.
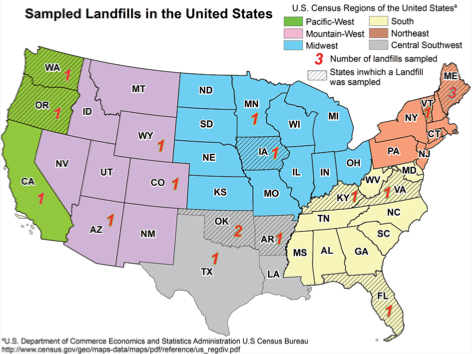

Figure 13. Map showing states where fresh (left) and final (right) leachate was sampled from 19 and 22 landfills from 2011 to 2012 (Masoner et al., 2014; Masoner et al., 2015)
Approximately 200 CECs were analyzed, including about 100 prescription pharmaceuticals, 30 industrial chemicals, 30 household chemicals (includes ten pesticides), 16 nonprescription pharmaceuticals, 16 steroid hormones, and 4 plant/animal sterols. These CECs were selected based on their quantity, persistence in the environment, and toxicological and ecotoxicological effects.
Prescription pharmaceuticals were most often detected, but household and industrial chemicals combined were observed more frequently and contributed over 80% of the total measured CEC concentration in both fresh and final leachate (Figure 15-16).

Figure 14. Frequency of contaminants of emerging concern (CEC) detection by chemical group (red bars) and the percent of total measured CEC concentrations by chemical group (blue bars) (Masoner et al., 2014)
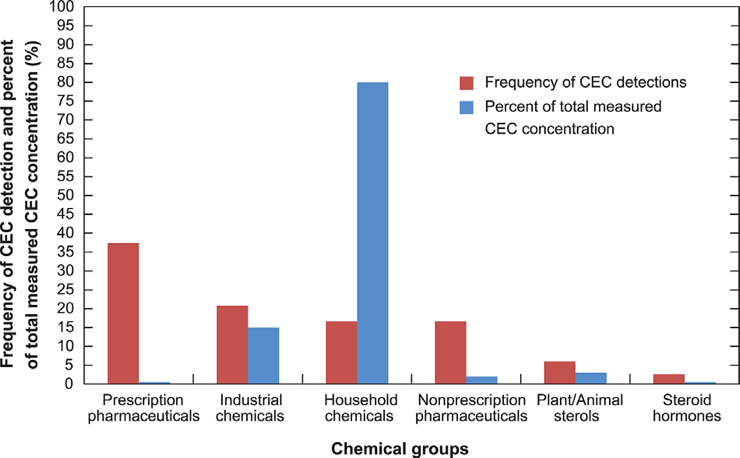
Figure 15. Frequency of contaminants of emerging concern (CEC) detection by chemical group (red bars) and the percent of total measured CEC concentrations by chemical group (blue bars) (Masoner et al., 2015)
The CECs content and frequency of detection in leachate is influenced as the following:
- Geochemistry
- Detection frequency of nine CECs increased as concentrations of non-volatile dissolved organic carbon (NVDOC) and inorganic analytes (Br-, B, K, Si, Co, Cr, and V) increased
- Landfill waste composition
- Addition of wastewater sludge
- Heterogeneous mixture of 70-80% municipal waste led to greater detection frequency and concentration of household chemicals
- Age of receiving waste
- Detection frequency: moderate age (11-20 yrs) > young age (4-11 yrs) ≈ old age (>20 yrs)
- Concentration: moderate age > young age > old age
- However, difference of detection frequency and concentration was not statistically significant, indicating recalcitrant nature of CECs
- Waste loading
- Moderate (0.125 – 0.5 million tons) and large (>0.5 million tons) waste loads had higher detection frequencies and concentrations compared to small waste load (<0.125 million tons)
- However, difference was not statistically significant, indicating another influencing factor
- Leachate production
- Detection frequency: large (>12 million gallons) ≈ moderate (5-12 million gallons) > small (<5 million gallons)
- Concentration: large (>12 million gallons) > moderate (5-12 million gallons) > small (<5 million gallons)
- However, difference was not statistically significant
- Precipitation
- Landfills in wet environments (>100 cm/yr) had greater detection frequency and concentrations than in drier area
- Difference in detection frequency and concentration of pharmaceuticals across wet to dry area was statistically significant
- Landfill status (active or closed)
- Concentrations were significantly greater in active sites than closed sites
- May be attributed to older leachate going through natural attenuation in closed landfills, smaller waste load in closed landfills, etc.
- Leachate treatment process (facultative lagoon, sequential batch reactor)
- Concentrations were significantly higher in untreated leachate that in treated leachate
- Leachate disposal method (WWTP, groundwater, irrigation, river)
- Concentration: WWTP > groundwater (table 00)
- Fresh vs final leachate
- CECs were detected less frequently and at lower concentrations in final leachate
Table 6. Ancillary landfill characteristics, total number of CEC detection, and total CEC concentrations in final leachate for the 22 landfills sampled in 2011 to 2012 (Masoner et al., 2015)
| Landfill | Landfill Type | Status | Leachate Treatment | Leachate Fate | Total CEC Detection | Total Measured CEC Concentration (ng/L) |
| LF1 | Municipal | Active | Facultative lagoon | River | 6 | 3,220 |
| LF2 | Municipal | Active | Facultative lagoon | WWTP | 40 | 6,308,430 |
| LF3 | Municipal | Active | Continuous, no treatment | WWTP | 58 | 5,029,820 |
| LF4 | Private | Active | Facultative lagoon | Irrigation | 14 | 43,980 |
| LF5 | Private | Active | Facultative lagoon | Irrigation | 19 | 65,900 |
| LF6 | Municipal | Closed | Continuous, no treatment | Groundwater | 9 | 40,490 |
| LF7 | Municipal | Closed | Continuous, no treatment | Groundwater | 19 | 46,900 |
| LF8 | Municipal | Active | Facultative lagoon | WWTP | 32 | 62,600 |
| LF9 | Private | Active | Facultative lagoon | WWTP | 20 | 65,200 |
| LF10 | Private | Active | Sequential batch reactor | WWTP | 17 | 17,280 |
| LF11 | Municipal | Closed | Continuous, no treatment | Groundwater | 12 | 23,550 |
| LF12 | Municipal | Active | Facultative lagoon | WWTP | 13 | 10,490 |
| LF13 | Municipal | Active | Facultative lagoon | WWTP | 36 | 534,500 |
| LF14 | Municipal | Closed | Continuous, no treatment | Groundwater | 25 | 200,120 |
| LF15 | Private | Active | Continuous, no treatment | WWPT | 45 | 20,722,770 |
| LF16 | Municipal | Closed | Continuous, no treatment | Groundwater | 2 | 7,660 |
| LF17 | Municipal | Closed | Continuous, no treatment | Groundwater | 1 | 380 |
| LF18 | Municipal | Active | Facultative lagoon | Irrigation | 37 | 195,470 |
| LF19 | Municipal | Active | Facultative lagoon | WWTP | 24 | 18,300 |
| LF20 | Municipal | Active | Facultative lagoon | WWTP | 29 | 24,660 |
| LF21 | Private | Active | Continuous, no treatment | WWTP | 40 | 1,774,980 |
| LF22 | Municipal | Active | Continuous, no treatment | WWTP | 37 | 622,010 |
The results of this research provide valuable information for future investigations of CECs in landfill leachate by discussing the factors influencing CEC species and concentration. Furthermore, the authors note that despite the fact that closed, unlined landfills and final leachate have lower detection frequencies and CECs concentrations compared to their respective counterparts, they are still of more concern due to the possibility of direct environmental contamination.
Conclusion
The objective of this report was to investigate landfill leachate treatment technologies suitable under different conditions. Leachate treatment technologies exhibit variable removal efficiencies for organic and inorganic contaminants. The concentration and types of contaminants in leachate are determined by a number of factors, including the age of the landfill. Biological treatment methods showed better performance when removing organic contaminants, whereas physicochemical treatment methods were more suitable for the removal of inorganic contaminants such as heavy metal. There were a few treatment technologies that were efficient in most circumstances, but required a trade-off with treatment cost. In addition to contaminant removal efficiency and cost, other factors such as on-site/off-site facility, need for leachate storage, required area, and local water standards must be considered to find the appropriate treatment method.
A part of this report also addressed a recent issue of CECs in leachate. CEC species and concentration in leachate were influenced by geochemistry, landfill waste composition, precipitation, landfill status (active/closed), leachate treatment process, leachate disposal method, and age of leachate.
The most important implication about landfill leachate from related studies is the need to continuously monitor and manage closed landfills to safely control leachate and prevent contamination of area surrounding the landfill. From this perspective, landfill leachate treatment is a crucial component of landfill management.
References
- Abbas, A. A., Jingsong, G., Ping, L. Z., Ya, P. Y., and Al-Rekabi, W. S. 2009. “Review on Landfill Leachate Treatments.” American Journal of Applied Sciences 6 (4):672-684.
- Agri-Food and Biosciences Institute. 2016. “On-Farm Small-Scale Anaerobic Digestion Event.” October 26, 2016. Accessed November 27, 2017. https://www.afbini.gov.uk/publications/farm-small-scale-anaerobic-digestion-event.
- Amokrane, A., Comel, C., and Veron, J. 1997. “Landfill leachates pretreatment by coagulation–flocculation.” Water Resources 31:2775–2782.
- Andreozzi, R., Caprio, V., Insola, A., and Marotta, R. 1999. “Advanced oxidation processes (AOP) for water purification and recovery.” Catalysis Today 53: 51–59.
- Berrueta, J. and Castrillon, L. 1992. “Anaerobic treatment of leachates in UASB reactors.” Journal of Chemical Technology and Biotechnology 54: 33–37.
- Beychok, M. 2010. “File: Air Stripping System.Png - Citizendium.” September 16, 2010. Accessed November 27, 2017. http://en.citizendium.org/wiki/File:Air_Stripping_System.png.
- Booth, S. D. J., Urfer, D., Pereira, G., and Cober, K. J. 1996. “Assessing the impact of landfill leachate on a Canadian wastewater treatment plant.” Water Environment Research 68(7):1179.
- Bove, D., Merello, S., Frumento, D., Arni, S. A., Aliakbarian, B., and Converti, A. 2015. “A Critical Review of Biological Processes and Technologies for Landfill Leachate Treatment.” Chemical Engineering & Technology 38(12):2115–2126.
- Canziani, R. and Cossu, R. 1989. “Landfill Hydrology and Leachate Production.” In Sanitary Landfilling: Process, Technology and Environmental Impact. Academic Press.
- Çeçen, F. and Aktaş, Ö. 2004. “Aerobic Co-Treatment of Landfill Leachate with Domestic Wastewater.” Environmental Engineering Science 21 (3):303-312.
- Christensen, T. H. and Kjeldsen, P. 1989. “Basic Biochemical Processes in Landfills.” In Sanitary Landfilling: Process, Technology and Environmental Impact. Academic Press.
- El-Fadela, M., Bou-Zeida, E., Chahineb, W., and Alaylic, B. 2002. “Temporal variation of leachate quality from pre-sorted and baled municipal solid waste with high organic and moisture content.” Waste Management 22:269–282.
- Foo, K. Y., Hameed, B. H. 2009. “An overview of landfill leachate treatment via activated carbon adsorption process.”Journal of Hazardous Materials 171: 54–60.
- Hassan, M., Zhao, Y., and Xie, B. 2016. “Employing TiO2 photocatalysis to deal with landfill leachate: Current statusand development.” Chemical Engineering Journal 285: 264–275.
- J&T Technologies, Inc. 2014. “JTEQUIP -ION Exchange.” Accessed November 27, 2017. http://www.jtequip.com/ion.html.
- Justina, M. Z. and Zupančič, M. 2009. “Combined purification and reuse of landfill leachate by constructed wetland and irrigation of grass and willows.” Desalination 246:157-168.
- KYDEP. 2012. “Membranes.” Kentucky Operator Certification Program (blog). March 14, 2012. https://kyocp.wordpress.com/2012/03/14/membranes/.
- Lin, S. H. and Chang, C. C. 2000. “Treatment of landfill leachate by combined electro-Fenton oxidation and sequencing batch reactor method.” Water Research 34: 4243–4249.
- Masoner, J. R., Kolpin, D. W., Furlong, E. T., Cozzarelli, I. M., Gray, J. L., and Schwab, E. A. 2014. “Contaminants of emerging concern in fresh leachate from landfills in the conterminous United States.” Environmental Science: Processes & Impacts 16:2335–2354.
- Masoner, J. R., Kolpin, D. W., Furlong, E. T., Cozzarelli, I. M., and Gray, J. L. 2016. “Landfill Leachate as a Mirror of Today’s Disposable Society: Pharmaceuticals and Other Contaminants of Emerging Concern in Final Leachate from Landfills in the Conterminous United States.” Environmental Toxicology and Chemistry 35 (4):906–18. https://doi.org/10.1002/etc.3219.
- Metcalf & Eddy / Aecom. 2014. “Wastewater Engineering: Treatment and Resource Recovery”, McGraw-Hill Education, Alexandria, Virginia, USA.
- Pokhrel, D. and Viraraghavan, T. 2000. “Leachate generation and treatment—a review.” Fresenius Environmental Bulletin 13: 223–232.
- Quasim, S. R. and Chiang, W. 1994. Sanitary Landfill Leachate Generation, Control & Treatment. Lancaster, PA: Technomic Publishing Company, Inc.
- Renou, S., Givaudan, J. G., Poulain, S., Dirassouyan, F., and Moulin, P. 2008. “Landfill leachate treatment: Review and opportunity.” Journal of Hazardous Materials 150:468–493.
- Schiopu, A.-M., Gavrilescu, M. 2010. “Options for the Treatment and Management of Municipal Landfill Leachate: Common and Specific Issues.” CLEAN – Soil, Air, Water 38 (12):1101–1110.
- Silva, A. C., Dezotti, M., and Sant’Anna, G. L. Jr. 2004. “Treatment and detoxification of a sanitary landfill leachate.” Chemosphere 55 (2): 207–214.
- Torretta, V., Ferronato, N., Katsoyiannis, I. A., Tolkou, A. K., Airoldi, M. 2017. “Novel and Conventional Technologies for Landfill Leachates Treatment: A Review.” Sustainability 9 (9).
- Townsend T.G., Powell J., Jain P., Xu Q., Tolaymat T., and Reinhart D. 2015. “Leachate Control, Storage, and Treatment.” In Sustainable Practices for Landfill Design and Operation. Waste Management Principles and Practice. Springer, New York, NY.
- US EPA, OSWER. 2016. “Bioreactor Landfills.” Overviews and Factsheets. US EPA. March 24, 2016. https://www.epa.gov/landfills/bioreactor-landfills.
- Zornberg, J. G., Jernigan, B. L., Sanglerat, T. R., and Cooley, B. H. 1999. “Retention of Free Liquids in Landfills Undergoing Vertical Expansion.” Journal of Geotechnical and Geoenvironmental Engineering 125 (7):583-594.
 Christina Kang
Christina Kang  Xiaohang Ji
Xiaohang Ji 





























4 COMMENTS
Eva*
Dec, 05, 2017 Hi there! In my opinion, the table 'Effectiveness of leachate treatment depending on landfill age (Abbas et al., 2009)' is the core of your report. Just wondering if it is updated and nothing new has come up since then! Christina Kang
Dec, 20, 2017 Eva,Thank you for the insightful comment. As you pointed out, the main point of our report is that leachate treatment technology can be selected partly based on the landfill age, or the biodegradation phase of the landfill. Indeed there are more effort now in creating a hybrid treatment solution composed of different treatment technologies to overcome the disadvantages of individual technologies. Therefore, the table constructed by Abbas et al. (2009) contain only the standalone, individual treatment technology that can be applied to landfill leachate of different age. The main purpose of this report was to introduce various treatment technologies, and it was not within scope to include all the various composite treatment technologies that are currently being studied. However, as you mentioned, significant advancement in composite treatment solutions was made since. Please let us know if you have additional comments.
Thank you,
Christina and Xiaohang
Edit Comment
Sally Simpson*
Dec, 11, 2017 Thank you for the interesting report. Can you provide some guidance on how many landfills process leachate on their own facility vs. sending it to a wastewater treatment facility? Christina Kang
Dec, 20, 2017 Sally Simpson,Thank you for the thoughtful request. Looking at the 22 representative landfills selected by Masoner et al. (2015) for their national study, 12 (54.5%) had on-site treatment facility, and of those 8 transferred treated leachate to a wastewater treatment plant (WWTP). 4 (18.2%) landfills had their leachate transported to a WWTP without any pre-treatment, and in 6 (27.3%) landfills, leachate was directly released into groundwater. These figures do not cover all of the landfills in the US, but it can be said that the landfills selected for the study by Masoner et al. (2015) are representative of the nation. Please let us know if you have additional comments.
Thank you,
Christina and Xiaohang
Edit Comment